IN VITRO
We use cell lines, 3D cell cultures and organoids for our basic research. These systems allow us to quickly and thoroughly address our MYC and statin research questions.
IN VIVO
We develop our own experimental models, and use established ones, to expand our in vitro work into more relevant systems. This is an essential step in translating our results into the clinic.
IN PATIENTS
Our ultimate goal is to positively impact patient outcome. We use our in vitro and in vivo data to inform clinical trial design, and work closely with multiple clinical teams to realize our goal.
MYC
Why do we study the MYC oncogene?
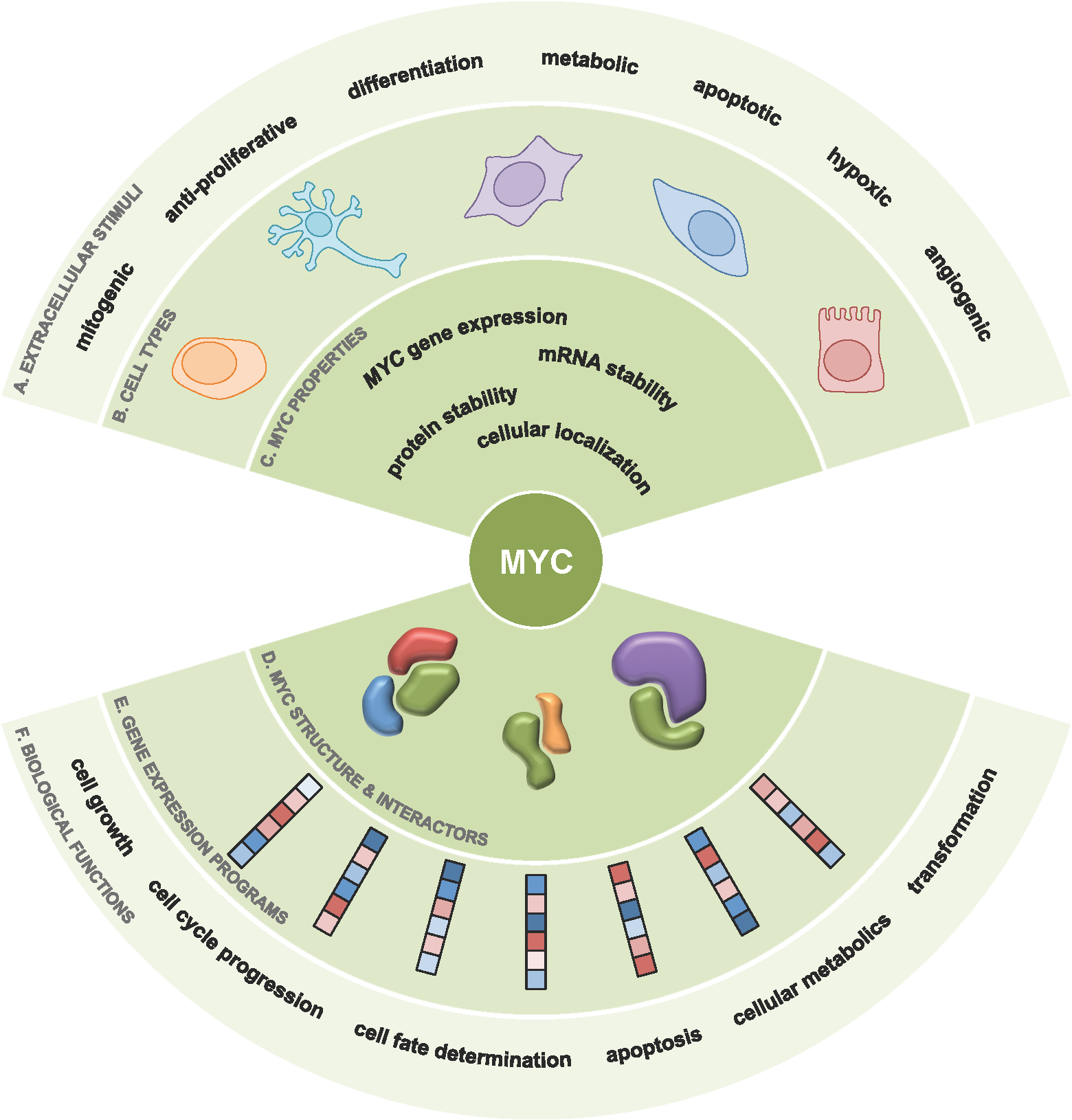
Expression of the MYC proto-oncogene is tightly controlled in normal cells, but when the MYC oncoprotein is deregulated and MYC activity unleashed, MYC drives the development of over 50% of human cancers. Our hypothesis is that by understanding the regulation and function of MYC in cancer, we can develop effective anti-MYC therapeutics. Such inhibitors of MYC would have an enormous impact on cancer patient care and outcome.
To date, there are no clinically viable therapeutics directly targeting MYC.
Traditional therapeutics rely on small molecular inhibitors to bind to enzymatic pockets or defined structures within its target. As MYC has no enzymatic activity and has very little defined structure, these approaches have not been fruitful, leading to the modern paradigm of MYC being “undruggable”.
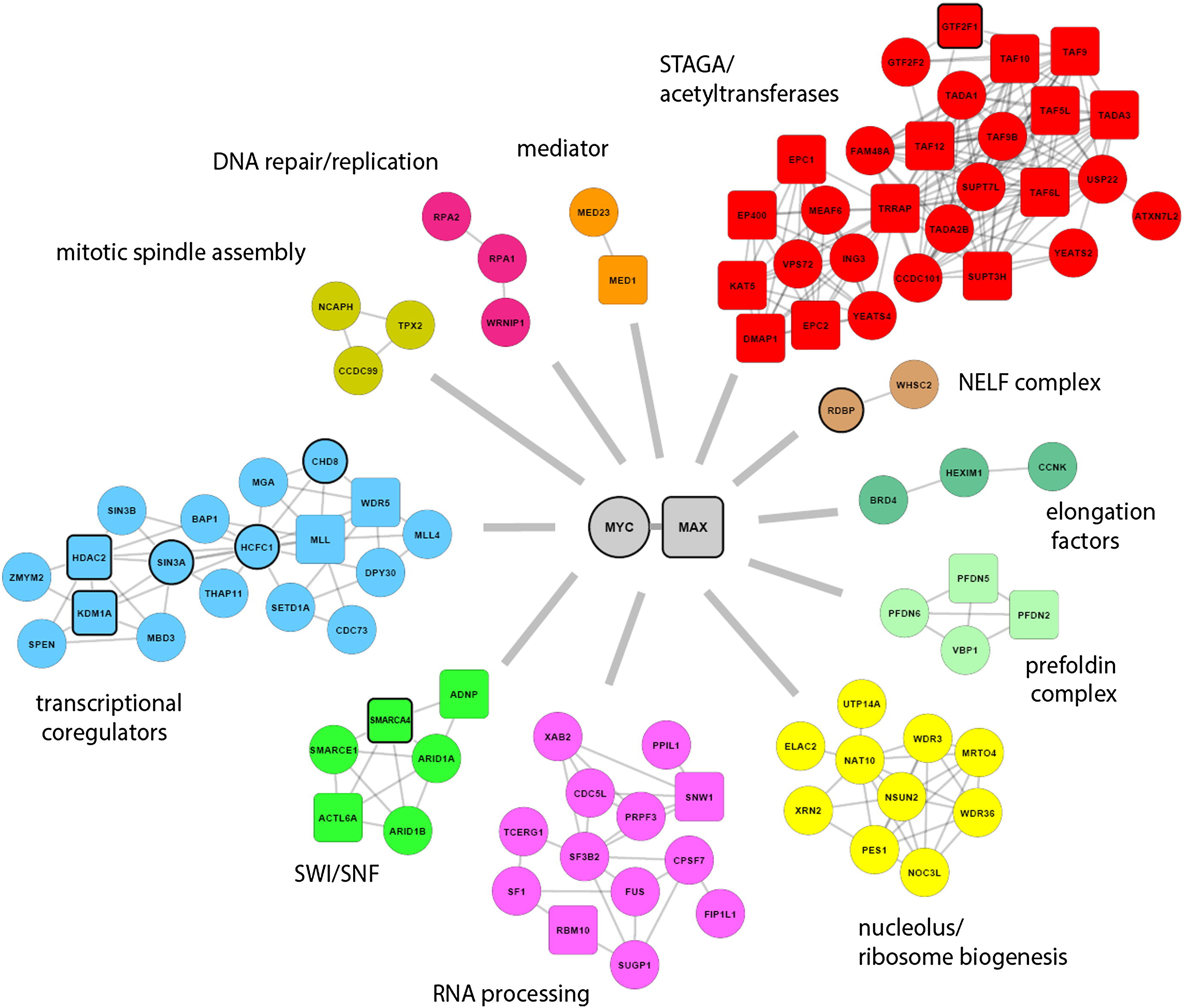
However this paradigm is shifting with the discovery of novel therapeutics inhibiting proteins within the larger network of MYC regulators and interactors. With the promise of developing indirect therapeutics against MYC, we are focusing on exploring how the MYC protein is regulated within the cell, a topic which is surprisingly poorly understood. We have been aided in this challenge with the advent of many promising new techniques such as BioID and CRISPR/Cas9 and leveraging this newfound understanding to identify novel therapeutic targets and strategies.
What are our important scientific questions?
- Which protein:protein interactors are critical to MYC function?
- How is the MYC protein regulated by post-translational modifications?
- Can we develop novel assays to score MYC-activity within tumour-like environments?
What are we doing to develop therapeutics against MYC?
We have used BioID, a mass spectrometry based approach to “tag” proximal proteins to MYC, which has dramatically expanded the MYC-interacome. This knowledge has identified hundreds of novel MYC interactors, whose function regulating the MYC protein or executing MYC’s tumorigenic activity is currently an area of focus. We have also identified novel and uncharacterized sites of post-translational modification on the MYC protein. Using a systematic approach and a number of novel in vitro and in vivo assays we have developed, we have scored the importance of these sites to the ability of MYC to drive tumourigenesis. We are using this new found knowledge, in combination with the larger MYC-interactome, to identify the proteins regulating these sites and determine the clinical benefit of targeting these proteins in Myc-driven cancers.
Our work aims to shift the MYC paradigm from “undruggable” by developing an arsenal of therapeutics against MYC, providing clinicians with the tools to dramatically improve patient care.
STATINS
Generating extra value from existing assets
Each year, the patents expire on numerous drugs, opening up possibilities to find new uses beyond their initial purpose. In an era of rising healthcare costs, finding new therapeutic uses for these drugs is a highly cost-effective and efficient route to better patient outcomes.
We have shown that statins, which are off-patent drugs widely used to lower cholesterol, also have potent anti-cancer effects in a variety of tumour types. Now we are attempting to identify which patients will respond to statins and determine how to improve these drugs’ anti-cancer effects. Our ultimate goal: translate these results into the clinic and improve patient care.
Identifying responders to statins
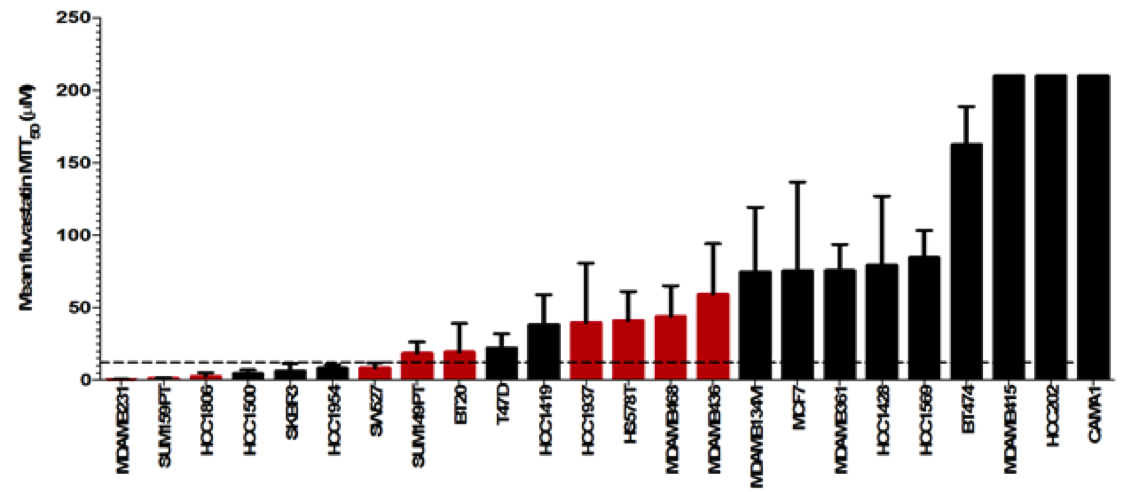
We have identified triple-negative breast cancer patients as a high-potential group for statin treatment. Currently, there are few targeted treatment options for these patients, and we believe that statins could aid survival. We are part of a large collaborative study that will fully characterize the response of different triple-negative breast cancers to statins, at the molecular, genetic and metabolic levels. This will allow us to identify patients who can benefit from these cheap, targeted drugs.
Improving the anti-cancer effects of statins
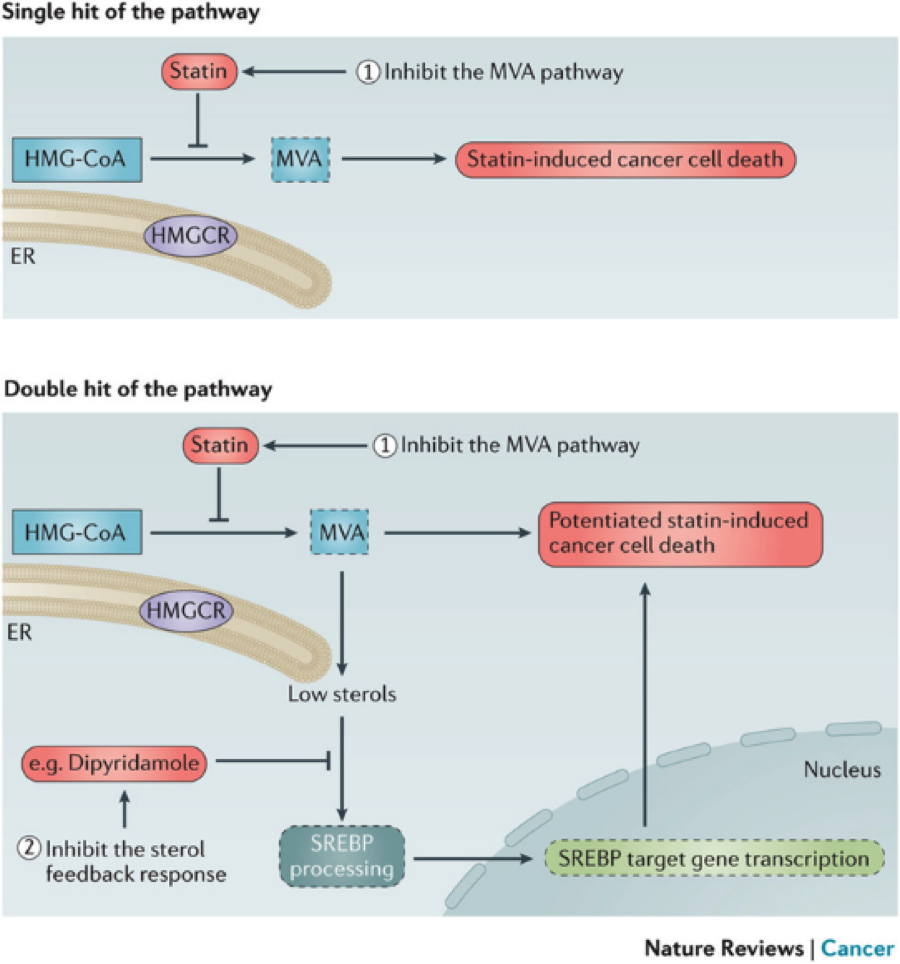
Most targeted therapies can induce a compensatory feedback or resistance mechanism in patients, leading to relapse. This can also happen with statins, which work by inhibiting the mevalonate pathway. Eventually, this leads to activation of the SREBPs (sterol response element binding proteins), which increase transcription of mevalonate pathway genes and restore it.
We aim to overcome this by targeting the SREBPs. We identified dipyridamole as a potent SREBP inhibitor, and have shown that combining this drug with statins can turn statin-insensitive cells into statin-sensitive one. It also increases the sensitivity of cells that already respond to statins alone. We are further characterizing the SREBPs and their regulatory pathways, with the aim to identify novel ways of inhibiting their activity and increasing the positive effects of statins in multiple cancer types.
Translating our research into the clinic
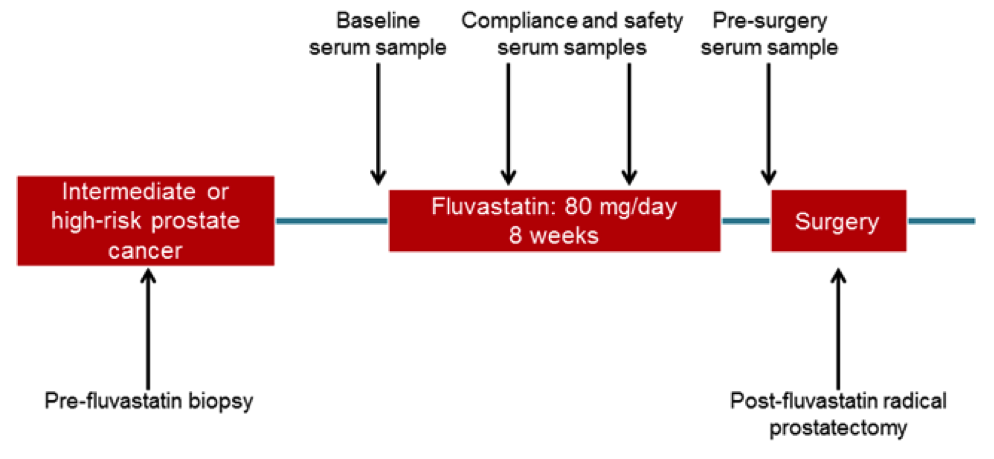
All our work is aimed at improving the survival and health of cancer patients. As a first step, we have led a clinical trial evaluating the effects of fluvastatin in high-grade prostate cancer. Patients were given normal doses of fluvastatin for six to eight weeks before they had their prostates surgically removed. We are currently evaluating markers of response, and aim to use this data to move forward with a larger randomized trial, and ultimately positively impact patient care.